Abstract
Background: Clonal hematopoiesis (CH) is defined by the acquisition of somatic mutations (MT) in hematopoietic stem cells with the propensity to expand over time. While CH is ubiquitous with aging, context-specific development of CH is heterogeneous and dependent on clonal pressures. Retrospective series have documented the role of CH in the development of therapy-related myeloid neoplasms (t-MN). In recent times, radioisotope-based therapies have advanced cancer management. Ibritumomab tiuxetan (Zevalin) is a radioimmunoconjugate approved for the treatment of relapsed low-grade follicular lymphoma (FL) and more recently, lutetium Dotatate has been used in neuroendocrine tumors and prostate cancer. Reports of accelerated t-MN with these therapies have generated concern. We studied a large cohort of relapsed FL patients treated with Zevalin to assess the prevalence and impact of CH.
Method: After IRB approval, we identified patients with FL treated with Zevalin who had stored DNA for CH assessment at any time point before or after therapy. DNA was extracted from mononuclear cells and subjected to a customized, targeted next-generation sequencing assay covering 220 CH-related genes (~ 1000x). Bone marrow (BM) sequencing was carried out in a subset at t-MN diagnosis. We compared clinical characteristics, MT patterns, variant allele fraction (VAF), and outcomes of those with and without t-MN. All statistical calculations were performed using R 4.0.1.
Results: Fifty-eight patients with relapsed FL were included, with a median age of 48.5 (IQR: 42-61.8) years, and 23 (40%) were females. The median follow-up duration was 17.8 (IQR: 15.7-21.9) years. At the last follow-up, 14 (24%) patients developed t-MN, and 23 (40%) deaths were documented. The median latency to t-MN after Zevalin exposure was 8.8 (IQR: 6.7-12.6) years, and the median overall survival (OS) after t-MN diagnosis was 0.9 (IQR: 0.24-2.2) years.
There were no significant differences in baseline demographics, FL stage, grade, FLIPI scores, and the number of prior therapies (median, 4 in both groups) between the t-MN vs. non-t-MN groups (all p>0.5; Table 1). At the time of Zevalin administration, all patients had a normal karyotype, and 46% (17/37) had prevalent CH, with 60% (35/58) having CH at any time point. In total, 97 variants were identified, 27 (28%) in t-MN and 70 (72%) in the non-t-MN group. Common variants seen in the t-MN group included PPM1D (n=7, 28%), DNMT3A (n=3, 11%), and TP53 (n=2, 8%), and in the non-t-MN group included DNMT3A (n=16, 23%), PPM1D (n=15, 21%), KMT2D (n=7, 10%), and TP53 (n=6, 9%).
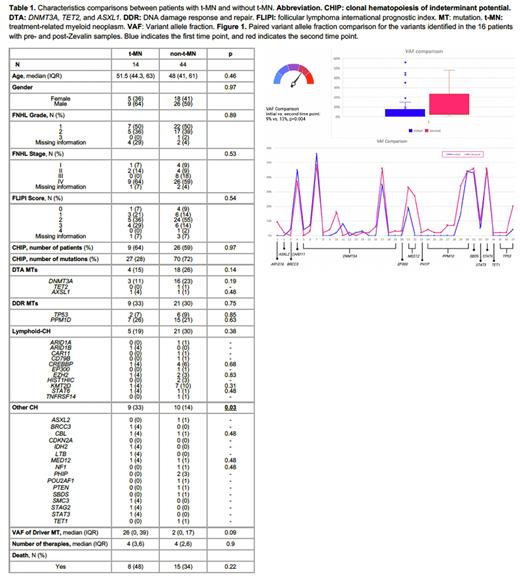
The prevalence of CH was not significantly different between the t-MN (64%) and non-t-MN (59%) groups (p=0.97). However, the t-MN group had a higher CH diversity (p=0.03), with a trend towards higher median VAF for driver MTs (26% vs. 2%, p=0.09) and a trend towards higher numbers of co-mutations (89% vs. 54%, p=0.06). The presence of CH prior to Zevalin therapy or post-therapy was not associated with a higher risk of t-MN (p=0.6 and 0.73, respectively). No other risk factors, including CH or biological CH groupings, were associated with t-MN.
Paired pre- and post- Zevalin exposure samples were available in 24 patients (37 MTs in 16 patients). Among 15 evaluable patients, 10 (67%) had CH at both time points and 5 (33%) had CH at the second time point only. The median annual change in VAF for DNMT3A (n=15) was 2% (range: -8% to 14%), and for DDR MTs (n=8) was 4.5% (range: 2% to 19%) (Figure 1). Four patients developed t-MN in this subset, 3 with CH and 1 without.
Thirteen of 14 evaluable patients who developed t-MN (7 each with t-MDS and t-AML) had an abnormal karyotype; 7 (50%) monosomal, 4 with chromosome 17 abnormalities. All 7 sequenced t-MN BM samples demonstrated new clones (n=15) at t-MN diagnosis. Common MT gained at t-MN transformation were TP53 (n=5, 33%), PPM1D and SETBP1 (both n=3, 20%), with 3 patients with biallelic TP53 inactivations.
Conclusion: We demonstrate the high prevalence of CH (60%) and t-MN (24%) in this heavily pretreated FL population who received Zevalin therapy. The spectrum of CH differed from age-related CH, with a high prevalence of MTs involving the DDR pathway, with no spliceosomal and a paucity of signaling MT, including at t-MN diagnosis. The median time between FL diagnosis to t-MN was 8 years and the mere presence of CH did not correlate with t-MN, highlighting the need to determine other mechanisms of clonal progression.
Disclosures
Mangaonkar:Bristol Myers Squibb: Research Funding. Ansell:SeaGen: Research Funding; Takeda: Research Funding; Bristol Myers Squibb: Research Funding; Regeneron: Research Funding; Affimed: Research Funding; Pfizer: Research Funding; ADC Therapeutics: Research Funding. Shah:Astellas: Research Funding; Celgene: Research Funding; Marker Therapeutics: Research Funding. Al-Kali:Astex: Other: research support to institution. Witzig:ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Curio Science: Honoraria; Karyopharm: Other: Clinical Trail Support; Kura Oncology: Other: Clinical Trail Support. Patnaik:Kura Oncology, Stemline Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal